FDA cleared pathways
510(k) summaries, UDI references, labeling history, and recall sweep notes prepared for biomed review.
Procurement, laboratory, imaging, and biomed teams use Hologic when they need IVD platforms, digital cytology support, mammography-related equipment programs, and home care device service wrapped in documented uptime processes.

510(k) summaries, UDI references, labeling history, and recall sweep notes prepared for biomed review.
Quality system documentation is organized around controlled service records, CAPA traceability, and supplier review cadence.
European deployment files include conformity references, IFU packs, and post-market surveillance routing.
Remote support workflows separate device telemetry from protected health information wherever integration allows.

Track-connected diagnostics, QC documentation, reagent lot control, and LIS routing.
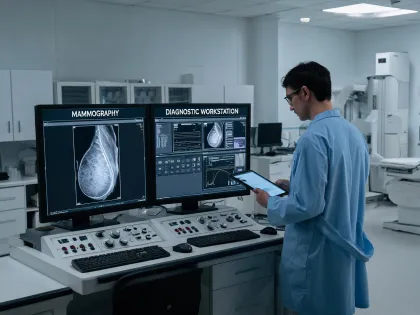
Digital mammography, detector support, preventive maintenance, and recall response planning.

Compact equipment packages with training, service contact routing, and onboarding checklists.

Device refresh support, caregiver education, asset labeling, and follow-up scheduling.
"The useful part was not another equipment quote. It was the field service plan, PM calendar, LIS integration checklist, and escalation map in one package."
"Hologic helped our biomed group standardize detector service records and train super users without interrupting a very full screening schedule."
"For home care assets, the training handoff mattered most. Caregivers had simple setup references, and our team had a repair route that did not disappear after delivery."